Ions are charged particles formed when atoms or molecules gain or lose electrons, altering their electrical charge. These charged entities play pivotal roles in various natural and synthetic processes, influencing chemical reactions, biological functions, and technological applications.
Atoms typically possess an equal number of protons and electrons, resulting in a neutral charge. When an atom gains or loses electrons, it becomes an ion with a net positive or negative charge. An atom that loses electrons becomes positively charged (cation), while an atom that gains electrons becomes negatively charged (anion).
Cations and anions exhibit distinct characteristics due to their differing charges. Cations are attracted to negatively charged regions while repelling other positive charges, whereas anions are drawn towards positive charges and repelled by negative charges. These properties influence their behavior in chemical reactions and their interactions with other ions or molecules.
Ions are fundamental in creating electrical conductivity in solutions. When ions dissolve in water or other solvents, they enable the flow of electricity as they move freely in solution, carrying charge. This phenomenon is crucial in biological systems, such as nerve impulses, where ions like sodium (Na+), potassium (K+), and chloride (Cl-) play essential roles in cellular signaling and muscle contraction.
Furthermore, ions are instrumental in the formation of ionic compounds through the attraction between oppositely charged ions. These compounds often exhibit unique properties like high melting points, electrical conductivity when dissolved, and crystal lattice structures due to the strong electrostatic forces between ions. Understanding the behavior and interactions of ions is fundamental in fields such as chemistry, biology, and material science, shaping our comprehension of numerous natural and artificial processes.
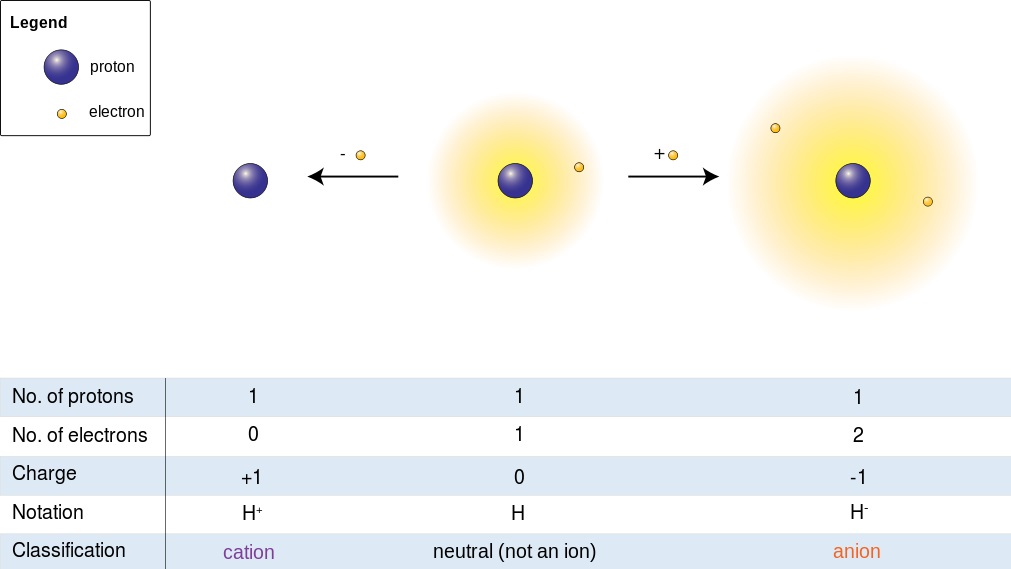
Defining cations and anions (Wikimedia)
Do you want to know more about ions? Let’s take a look at these 30 interesting facts about ions.
- Electrical Charge: Ions are charged particles resulting from atoms gaining or losing electrons.
- Cations and Anions: Cations have a positive charge due to losing electrons, while anions have a negative charge from gaining electrons.
- Neutralization: Oppositely charged ions can neutralize each other, forming electrically neutral compounds.
- Ionic Bonds: Ions with opposite charges form ionic bonds, creating compounds like table salt (NaCl) or calcium carbonate (CaCO3).
- Electrolytes: Ions in solution enable electrical conductivity, crucial for nerve function and muscle contraction in the body.
- Metallic Ions: Some ions, like iron (Fe2+ and Fe3+), copper (Cu2+), and zinc (Zn2+), are essential for various biological processes.
- Polyatomic Ions: These ions consist of multiple atoms and possess a net charge, like the sulfate ion (SO4^2-) or ammonium ion (NH4+).
- Hydrogen Ions: Protons in aqueous solutions are often referred to as hydrogen ions (H+).
- Ion Channels: These specialized proteins facilitate the movement of ions across cell membranes, regulating cell function.
- Ionic Liquids: These are liquid salts composed of ions and have unique properties useful in various industrial applications.
- Ionosphere: This region of the Earth’s atmosphere contains ions formed by solar radiation and influences radio wave propagation.
- Ionizing Radiation: High-energy particles or waves, like alpha, beta, and gamma rays, can create ions in substances they interact with.
- Ion Mobility Spectrometry: A technique using electric fields to separate and detect ions based on their mobility in a gas.
- Ion Thrusters: These propulsion systems in spacecraft use ions to generate thrust, allowing for efficient and precise maneuvering.
- Ion Exchange Resins: These materials can selectively exchange ions in solutions, used in water purification and chemical separations.
- Mass Spectrometry: Analytical technique identifying substances by measuring the mass-to-charge ratio of ions.
- Ion Selective Electrodes: These devices measure ion concentrations in solutions, valuable in medical and environmental monitoring.
- Eutectic Ionic Liquids: Mixtures with extremely low freezing points, potentially useful as environmentally friendly solvents.
- Ion Implantation: A technique introducing ions into materials for altering their properties, commonly used in semiconductor manufacturing.
- Isotopic Ions: Different isotopes of an element can form ions with slightly varied masses due to differing numbers of neutrons.
- Ozone Formation: Ozone in the Earth’s stratosphere is formed by the interaction of oxygen molecules and ultraviolet radiation, producing ions.
- Ion Cyclotron Resonance: A method in nuclear physics using magnetic fields to manipulate and study ions’ behavior.
- Ion Laser: These lasers generate beams of ions, useful in scientific research and industrial applications.
- Ion Bombardment: The process of bombarding a surface with ions, altering its properties or cleaning it.
- Ionic Crystals: These crystals exhibit distinct cleavage patterns due to their structure, commonly observed in minerals.
- Ion Detectors: Devices capable of detecting and measuring ions, essential in various scientific instruments.
- Ion-Exchange Chromatography: A technique separating ions based on their interactions with a stationary phase, widely used in analytical chemistry.
- Electron Capture: A process in which a nucleus captures an electron, forming a lower energy state and generating an ion.
- Photoionization: The process of ejecting an electron from an atom or molecule by absorbing photons, creating ions.
- Astrophysical Ions: Ions in interstellar space and cosmic environments play a crucial role in understanding celestial bodies’ composition and behavior.
Ions, these charged particles resulting from atoms gaining or losing electrons, are ubiquitous and influential across numerous scientific disciplines and everyday applications. Their unique properties and interactions are integral to understanding chemical reactions, biological processes, and the behavior of matter on both macroscopic and microscopic scales. From their pivotal role in forming compounds and facilitating electrical conductivity to their applications in various technologies and analytical methods, ions represent a fundamental aspect of our understanding of the natural world. Studying ions continues to unveil new insights into the intricate workings of chemistry, physics, and biology, shaping advancements in fields as diverse as medicine, materials science, and space exploration.





